At BeautyAnswered, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What is a Carbomer?
A carbomer is a homopolymer of acrylic acid, which is cross-linked, or bonded, with any of several polyalcohol allyl ethers. Usually appearing as a white powder, the compound is used as a thickener and emulsion stabilizer. Best known for its use in the cosmetic industry, it also has practical applications in medicine and hygiene. Many agencies consider the various types to be perfectly safe, although some of the substances used to neutralize their pH can be problematic.
Chemical and Physical Traits

Similar to other polymers, carbomers are made of long chains of many smaller, repeating molecules, which have a large number of bonds. Although the molecular weight varies based on the exact molecules found in the chain, it typically is relatively high. These compounds are capable of absorbing large amounts of water, increasing in volume up to 1,000 times in some cases, so they can form gels and thick solutions that are stable and resistant to spoilage. Specifically, it's the polyalcohol portion that gives a carbomer its high water solubility, unusual in polymers of this size. In general, they are white powders when not added to a solution.
Labeling

Scientists are able to make different types of carbomers, each of which has a slightly different molecular structure. To keep these different kinds straight, they use a numerical suffix and capitalize the word as in a proper title or name, such as Carbomer 940. Under this labeling system, the number indicates the average molecular weight of the polymer chains.
Applications

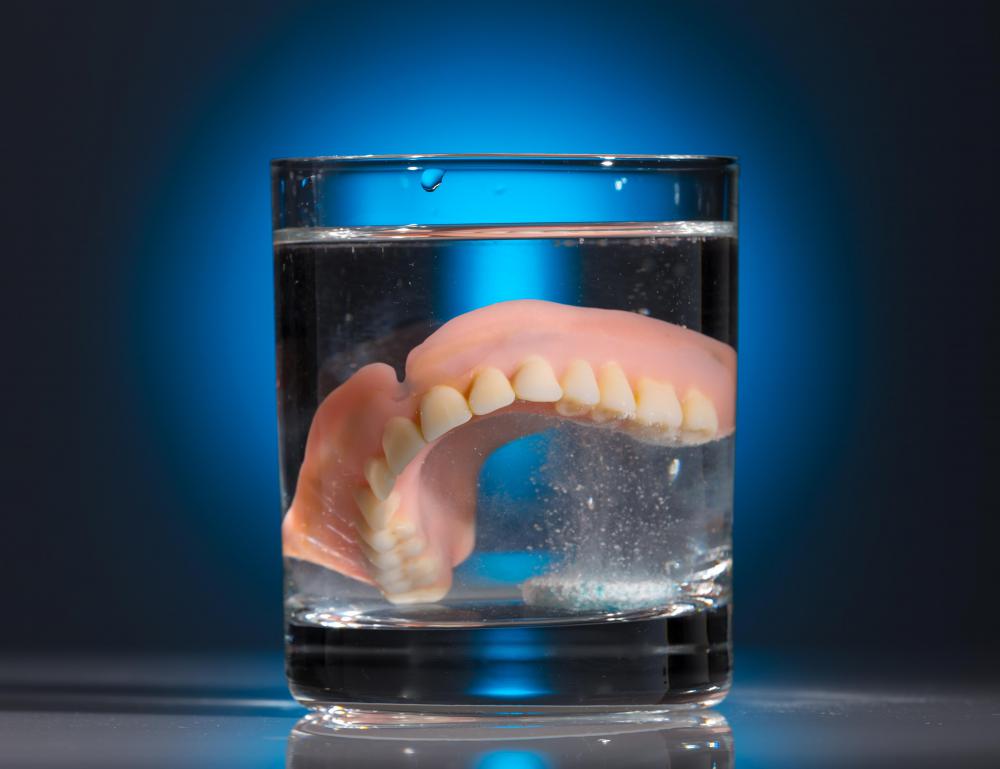
Most commonly, companies use carbomers in cosmetics — in fact, they were created specifically for this industry. They have a number of hygienic uses as well, however. They are used in toothpastes and gels, for example, as well as in denture cleansers, and they are common in shampoo. Many topical creams and lotions, such as those used to keep skin hydrated or treat various skin conditions, use them as well, and they also are found in certain types of eye drops. Another familiar product that uses them is disposable diapers — it's these compounds that absorb the water in the urine and make the diaper swell when wet.
Reasons for Use

The consistency and texture of cosmetics and personal care products are of critical importance to the consumer, and different agents are routinely added to these products to control the tactile qualities. Natural plant gums or extracts have been used, but these are often less than ideal since they can vary in quality from batch to batch. They may also cause allergic reactions in sensitive users and are often subject to bacterial attack and spoilage, requiring the addition of antibacterials.

By contrast, a carbomer is an excellent thickening agent, is consistent from batch to batch, will not support bacterial growth and is hypo-allergenic. It also has a particularly nice "skin feel," producing solutions and gels that feel rich and luxurious to the touch. As an emulsion stabilizer, carbomers keep oils or creams suspended in water and prevent separation.

The size of these polymers and the way they are bonded lets them serve as net-like structures in water, allowing them to support fine, insoluble particles better than biologically-derived gels, and at lower concentrations. For this reason, people often look them as suspension agents when they need to disperse fine solids in creams or lotions.
Safety
In general, regulatory agencies consider carbomers to be safe, and they have a use history that spans at least five decades. They are included in the United States Pharmacopeia, and Carbomer 934P/974 is approved for use in oral formulations and ophthalmic solutions, an indication of its low toxicity. The Environmental Working Group (EWG) also reports that they are not a concern in terms of bioaccumulation or a hazard to the environment. When they are full strength, however, they do have the potential to cause some irritation to the eyes and skin, so in most cases, manufacturers adjust their concentration.

Aqueous solutions of carbomer are mildly acidic, usually having a pH of about 3. Companies typically adjust the acidity of products containing these compounds with a base, such as triethanolamine, to bring it up to a more neutral value. Although the polymers themselves generally are not problematic if manufacturers adjust their strength appropriately, the substances used for neutralizing often have their own set of safety concerns. Reading the label does not always alert consumers to the presence of these neutralizers, depending on their classification and regulation.
AS FEATURED ON:
AS FEATURED ON:
















Discussion Comments
Very good info. Thanks. However, this carbomer homopolymer is kind of scary. It's also used in a number of OTC vaginal lubricants and moisturizers. I can't imagine using anything for delicate lady parts with the words "poly-alcohol or allyl ethers" in it. After 20 years of menopause, I recommend an organic product like Aloe Cadabra. It's a 2-in-1 lube and moisturizer and it's edible! No parabens either.
Why do they put it in a tube of 'tooth-whitening' gel?
How much carbomer is needed for Alkali Oxide or amine to neutralize it?
Post your comments