At TheHealthBoard, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What is GLUT4?
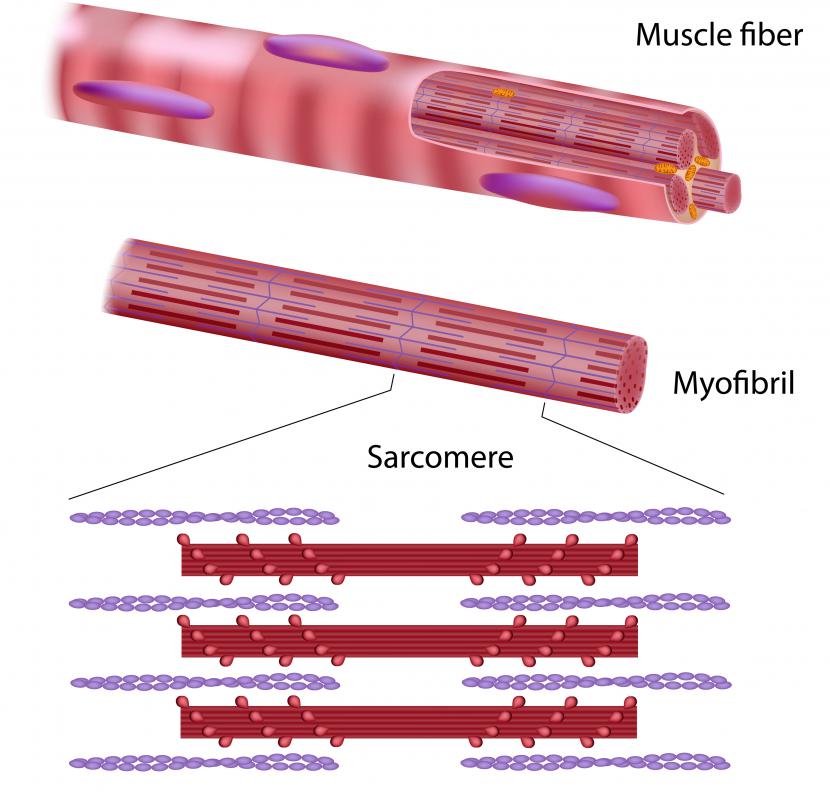
GLUT4 is a glucose transport protein found in fat and striated muscle cells. In a person with normal metabolism, insulin is secreted from the pancreas after eating. Insulin signals fat and muscle cells to absorb glucose from the blood by binding to the insulin receptor on the surface of fat and muscle cells.
When an insulin receptor is activated, it induces the GLUT4 protein to move from reserves held inside cells. This protein can also be recruited to the cell surface through muscle contraction. In the absence of insulin or muscle contraction, it is stored in vesicles within the cell.

When GLUT4 is at the cell surface, glucose is transported into the cell down its concentration gradient in a process called facilitated diffusion. A phosphate group is then immediately added to the glucose by the enzyme hexokinase in a process called phosphorylation, creating glucose-6-phosphate, which cannot diffuse outside the cell. Glucose-6-phosphate can be used as an energy source when it is metabolized through glycolysis and the Krebs cycle.

Defects in the activity of this protein have been implicated in some forms of insulin resistance or pre-Type 2 diabetes. Insulin resistance is a condition where the pancreas secretes insulin, but fat and muscle cells do not respond to it by taking up glucose. The pancreas compensates by secreting extra insulin to help cells take up glucose, and insulin-resistant patients often have high levels of glucose as well as a high blood insulin concentrations. The pancreas eventually fails to keep up with the body's need for insulin, and Type 2 diabetes can result. The role of GLUT4 in insulin resistance is an active area of diabetes research.

Insulin triggers the transport of this protein to the cell surface through a complicated signal transduction mechanism involving the phosphorylation and dephosphorylation of proteins and lipids. Following insulin binding, the insulin receptor phosphorylates a protein called Insulin Receptor Substrate 1 (IRS-1). IRS-1 works through lipids and other proteins to move GLUT4 to the cell surface. When blood insulin levels decrease, GLUT4 must be removed from the cell surface in order to slow glucose uptake into the cell. This process is less well studied.

GLUT4 is a member of a family of glucose transport proteins. Glucose transporters are integral membrane proteins containing 12 membrane-spanning helix domains. This protein is the only member of the family that is regulated by insulin. While it is specific to muscle and fat tissue, other members of the glucose transporter family are specific to other tissues. For example, GLUT3, a high-affinity glucose transporter, is the primary glucose transporter in neurons.
AS FEATURED ON:
AS FEATURED ON:














Discussion Comments
I have a bio test coming up soon. This clears up so much!
Post your comments